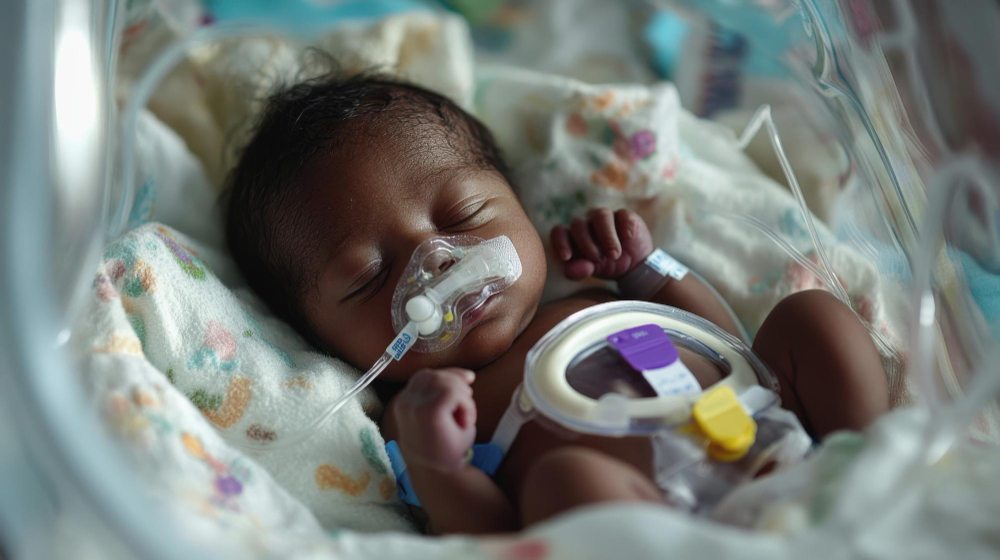
Carried silently by millions of women, Group B Strep can turn a baby’s first 90 days into a fight for survival, and scientists are now in the final stages of developing a vaccine to close this deadly gap between mother and child.
The first three months of a baby’s life are the most dangerous. For Group B Streptococcus (GBS), a bacterium carried naturally by millions of women worldwide, those 90 days represent a window of vulnerability that researchers and clinicians are racing to close.
“When you hear us talk about ‘infant GBS disease burden,’ this is what we mean,” says Dr Stephanie Schrag, who has spent 25 years leading GBS and neonatal sepsis programmes at the U.S. Centers for Disease Control and Prevention. “We’re talking about babies in their first days and weeks of life who develop sepsis, pneumonia, or meningitis from an infection that is entirely preventable.”
GBS colonises the vagina, rectum, urinary tract, upper respiratory tract, and gastrointestinal tract of healthy adults, most often without symptoms. For pregnant women, this colonisation poses no threat to their own health. But for their newborns, it can be deadly.
Infants are among those at highest risk for GBS infection, alongside pregnant women themselves, older adults over 65, and people with compromised immunity or chronic conditions like diabetes, cancer, and HIV.
Nairobi conference offers clearer picture what it would take to protect newborns
Infection occurs when the bacteria spread from colonised areas into normally sterile parts of the body like the blood, lungs and spinal fluid.
A major study presented at the International Symposium on Streptococcus agalactiae Disease (ISSAD) that ended this week in Nairobi, Kenya, offers the clearest picture yet of what it would take to protect newborns.
Dr Schrag and her CDC team conducted a large natural history study spanning 12 years and eight surveillance sites, comparing infants under 90 days who developed invasive GBS disease with those who did not – all born to mothers who were colonised with the bacteria.
Using dried blood spots collected from routine newborn screening, researchers measured anti-capsular polysaccharide IgG antibody concentrations (immune proteins produced by the body to fight bacteria) in babies’ blood at birth. What they found confirms what many suspected: antibodies matter, and the levels needed for protection vary by both serotype and timing of disease.
For serotype III – the most common cause of infant GBS disease globally – a baby needs an antibody concentration of 0.27 micrograms per millilitre at birth to achieve 75 per cent protection against early-onset disease, which occurs in the first week of life. For 90 per cent protection, that threshold jumps to 4.0 micrograms per millilitre.
Science of protection now being translated into science of prevention
For late-onset disease, striking between one week and three months of age, the thresholds are lower: 0.03 to 0.09 micrograms per millilitre for serotype III. But those antibodies must persist in the baby’s bloodstream for weeks as the infant’s own immune system matures.
“There was no vaccine used in this study,” Dr Schrag emphasises. “It’s a natural history study. The controls had some opportunity to get GBS disease. We are measuring the antibody concentrations that distinguish babies who got sick from those who didn’t.”
The science of protection is now being translated into the science of prevention. Pfizer is currently conducting a Phase 3 trial of a six-valent GBS vaccine designed to protect against the six most common disease-causing serotypes. The study, named BEATRIX, is enrolling healthy pregnant participants across 18 countries, with representation of both high-income and low- and middle-income settings.
“The proposed indication is to administer this vaccine to pregnant women to protect infants from all the diseases you’ve been hearing about – sepsis, pneumonia, meningitis – against both early-onset and late-onset disease,” explains Dr Barbara Pahud, clinical development lead for the GBS programme at Pfizer.
Vaccine generates antibodies that transfer successfully to infants
The vaccine uses 20 micrograms per serotype without adjuvant, for a total dose of 120 micrograms per half-millilitre. Earlier phase studies demonstrated an acceptable safety profile, with most adverse events mild or moderate – similar to other vaccines administered during pregnancy.
Importantly, the vaccine generates antibodies that transfer successfully to infants, reaching levels in newborns associated with reduced risk of invasive GBS disease. Transplacental transfer ratios vary by serotype but range from 0.55 to 0.74 for the final dose selected.
A booster dose study showed that antibody levels remain higher than baseline even two years after vaccination, and that a second dose is well-tolerated and elicits immune responses consistently higher than after the first dose.
The Phase 3 trial is not designed as a traditional efficacy study – proving the vaccine prevents disease would require enrolling between 50,000 and 100,000 pregnant women, a logistical challenge given the variability of serotype incidence across regions. Instead, regulators have accepted an immunogenicity endpoint: if vaccinated mothers achieve antibody levels associated with protection, the vaccine can be licensed on that evidence, with Phase 4 studies to follow after introduction.
In Argentina, maternal RSV vaccine was approved in 2023
While the world awaits a GBS vaccine, another maternal immunisation programme offers a glimpse of what is possible. In Argentina, a maternal RSV vaccine was approved by regulators in September 2023, recommended by the national immunisation advisory group in November, included in the national schedule in December, and first administered in March 2024.
Coverage reached 67.8 per cent in the first season, with over 147,000 doses administered. Real-world effectiveness studies confirmed the vaccine worked: a 78.6 per cent reduction in RSV hospitalisations in infants up to three months old, and 71.3 per cent up to six months.
Dr Gonzalo Pérez Marc, who led the Argentine effort, identified three pillars of success: clear national policy, operational access, and effective communication. “Provinces with higher coverage were those that implemented more aggressive and sustained communication campaigns targeting both healthcare professionals and the general population,” he explains.
The first 90 days of life are a window of vulnerability — a period when a common bacterium carried by healthy mothers can become a killer of newborns. But they are also a window of opportunity.
“We have the science,” says Dr Schrag. “We know the antibody levels needed for protection. We know which serotypes cause disease. We have vaccines in development. The question now is not whether we can prevent GBS, but whether we can deliver prevention to everyone who needs it.”