While classmates and neighbours succumbed to kala-azar, Luke Kanyang’areng’ cheated death—after a month of needle-stung mornings, each injection burning away the parasite… and a piece of his childhood.
At 14, Luke Kanyang’areng’ fell sick with kala-azar (visceral leishmaniasis). At first, he had a constant fever, headaches, and no appetite—but it took days before doctors confirmed what was wrong.
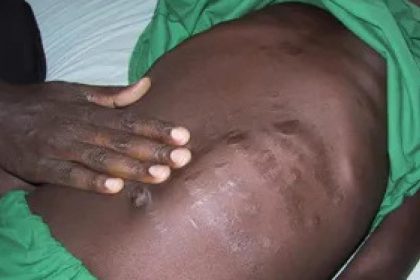
Back then, West Pokot had few labs to test for kala-azar. Luke had to travel more than 60 kilometres to Kitale just to get diagnosed. His case was tricky because, unlike most patients, he didn’t have a swollen spleen or belly—the usual signs of the disease.

Luke says the situation has changed as today, “we have several hospitals fully equipped with laboratories. We don’t have to travel as far as Kitale to get tested.”
Unlike many classmates and neighbours who died from kala-azar, Luke survived—thanks to timely treatment. But the cure came at a cost: 30 straight days of painful injections, so dreaded that many patients avoided hospitals altogether.
Luke’s battle with kala-azar didn’t just save his life—it changed his path forever. Today, he works as a nurse at Kanyarkwat Health Centre in West Pokot, where he’s seen real progress: hospitals like Kacheliba Sub-County Hospital now lead the fight against the disease with better equipment and research.
The word Kala-azar refers to the greyish or blackish discolouration of the skin during infection. It is a combination of the Hindi word for black (kala) and the Persian word for disease (azar).
Kala-azar is caused by parasites called Leishmania, with over 20 different types. The deadliest form is Visceral Leishmaniasis (kala-azar), which kills over 95% of untreated patients. Most cases occur in Brazil, East Africa and India, with 50,000-90,000 new infections yearly – but only 25-45% get reported, according to the World Health Organization (WHO).
There are two other types: Skin Leishmaniasis, which causes sores on exposed skin (600,000-1 million cases/year in tropical regions) and Mucosal Leishmaniasis, which destroys nose and mouth tissue (found in South America and Africa). The disease spreads through sandfly bites. About 70 animal species (including humans) can carry these parasites.
Dr Paul Kibati, the Technical Officer for Tropical Neglected Diseases at AMREF Health Africa, warns that children are particularly vulnerable to Kala-azar due to their developing immune systems.

“In areas with cases among children, malnutrition is also a very big factor because malnourished children have a weaker immune system, thus more likely to contract the disease,” explains Dr Kibati.
Children are also more exposed to sandflies as they spend significant time around their breeding sites like houses, riverbanks and acacia tree barks common in pastoral areas.
Dr Kibati clarifies that being bitten by sandflies doesn’t immediately mean infection as the body can fight back kala-azar “but in children whose immunity system is still developing, fighting the infection may be compromised.”
Symptoms of Kala-azar include; swollen spleen, abdomen, and liver, persistent fever and weight loss. The parasite enters the lymphatic system and replicates within white blood cells, a situation that can lead to liver and kidney failure.
“This is why there is enlargement of the spleen because they are proliferating within the immune system,” explains Dr Kibati. “This affects the cell-line for blood production which is why anaemia is very common among Kala-azar patients.”
Platelet count and white blood cell count also drop, leading to bleeding tendencies and compromised immunity.
Dr. Kibati stresses that blood transfusions are crucial for treating kala-azar, but currently, treatment is only available at specialized hospitals that can monitor liver and kidney health and provide transfusions. There is, therefore, need for access to blood and blood products for patients with Kala-azar,” he says, noting that the aggressive treatment regimen for Kala-azar requires hospitalization.
Dr Cherinet Bayuh, the Senior Market Access Manager at Drugs for Neglected Diseases Initiative (DNDi) Eastern Africa, explains that while hospitalization is necessary for Kala-azar patients, the initial aggressive treatment regimen poses many challenges.

Use of sodium stibogluconate as a monotherapy – the main drug used to treat Leishmaniasis – is no longer the preferred first-line treatment due to toxicity.
Following clinical trials by DNDi and other partners, WHO recommended a different regimen as the first-line treatment for uncomplicated cases. The regimen contains sodium stibogluconate with paromomycin given intramuscularly for 17 days. This new regimen has been received well, and hospitalization has been reduced from 30 to 17 days.
However, patients still have to deal with side effects such as injection site pain and systemic toxicity affecting internal organs like the pancreas and liver, which may also cause life-threating toxicity to the heart. DNDi has conducted trials on oral Miltefosine which, while less toxic, is currently only approved for HIV-Kala-azar co-infection, primarily in Ethiopia.
“The Kala-azar patients who present very late with severe co-infections and complications do not have good treatment outcomes,” Dr Bayuh notes, adding that post-Kala-azar Dermal Leishmaniasis (PKDL), a skin rash complication, may be reported during or after treatment and is more common in Sudan than Kenya or Uganda.
For some PKDL patients, the lesions may disappear by themselves, but for some, they will progress and require treatment. He says these patients are potential transmitters of the disease and should utilize treatment.
WHO aims to eliminate Visceral leishmaniasis as a public health problem defined as less that 1% case fatality rate due to primary visceral leishmaniasis by 2030 globally while in eastern Africa the targets are 90:90:100 fewer than 1,500 new cases reported per year by 2030, about 90 percent reduction from the 13-year average including 25 % underreporting in 2010-2022. This can be broken down to 90 percent of VL cases detected and started on treatment within 30 days of onset of symptoms by 2030, 100% decline in VL deaths in children by 2030, and 100% of VL-HIV co-infected patients started on antiretroviral therapy and referred to HIV clinics and all PKDL cases detected, reported and managed.
Dr Mutono Nyamai, a senior research fellow at the Centre for Epidemiological Modelling and Analysis (CEMA), says they’re using statistical and mathematical methods to map out where the burden of disease is, to guide intervention strategies.

CEMA’s models help policymakers understand the potential impact of different interventions and make informed decisions about resource allocation. So far, Bangladesh is the only country validated by WHO for eliminating Kala-azar.
Dr Kibati attributes Bangladesh’s success to effective vector control, community engagement, and active case finding.
“They mapped where the vectors were, either breeding indoors or outdoors, and employed a huge community workforce to spray in those communities, unlike Kenya which is high in active case management and treatment,” Dr Kibati says.
This was coupled with widespread community sensitization and targeted engagement in rural areas, enabling the success of vector control experts. He suggests that Kenya could benefit from targeted vector control due to its vast arid and semi-arid regions.
AMREF works with the Ministry of Health at both the national and county levels to implement its annual Kala-azar work plan. This includes treating patients, improving reporting systems, and training healthcare workers and community health promoters.
Dr Kibati says that previously, cases were reported through a WHO system, which gave minimal visibility to the in-country leadership, especially senior officials. “So we are mainstreaming this back to the Ministry of Health just to ensure that visibility can be created.”
Already, 12 Kenyan counties are endemic for Kala-azar: Mandera, Wajir, Garissa, Kitui, Isiolo, Marsabit, Turkana, West Pokot, Baringo, Kajiado and Tharaka Nithi, with climate change contributing to the spread of the disease to new areas such as Samburu.